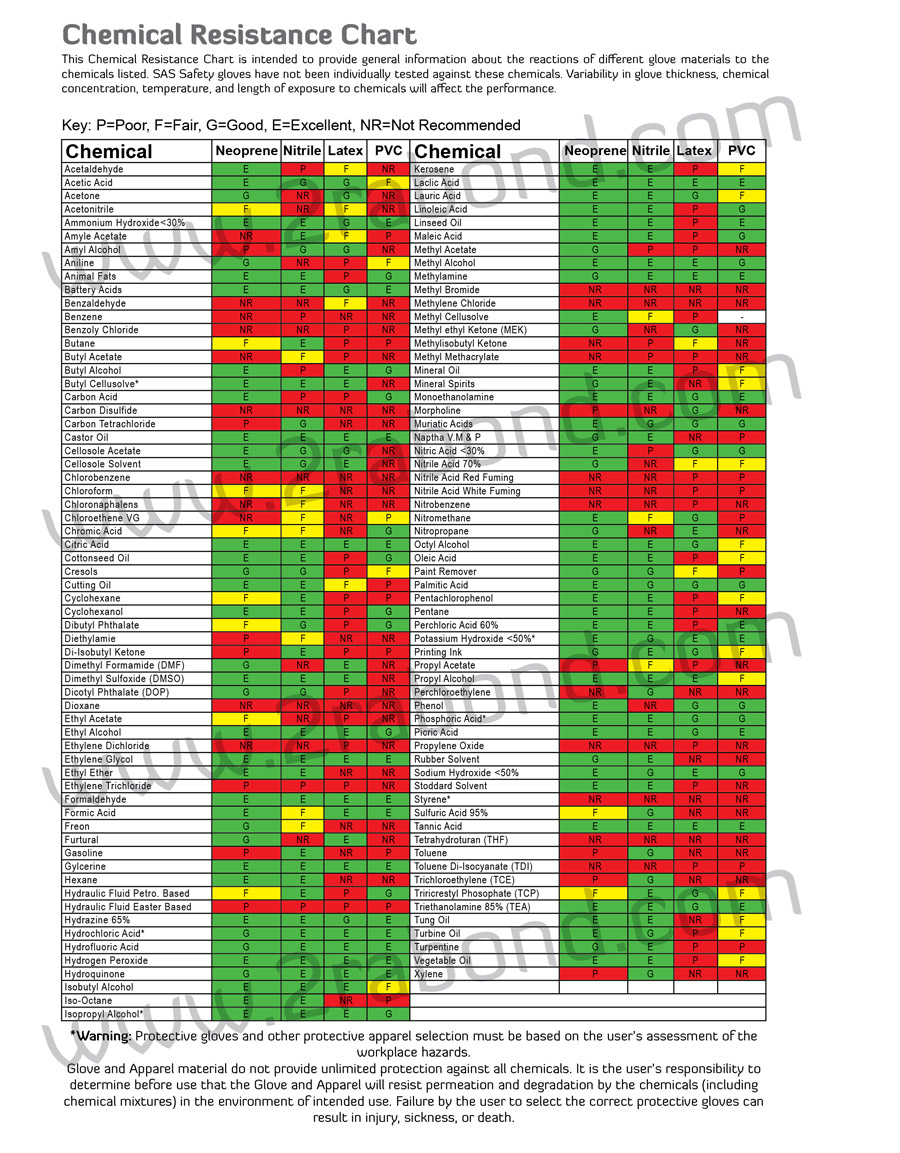
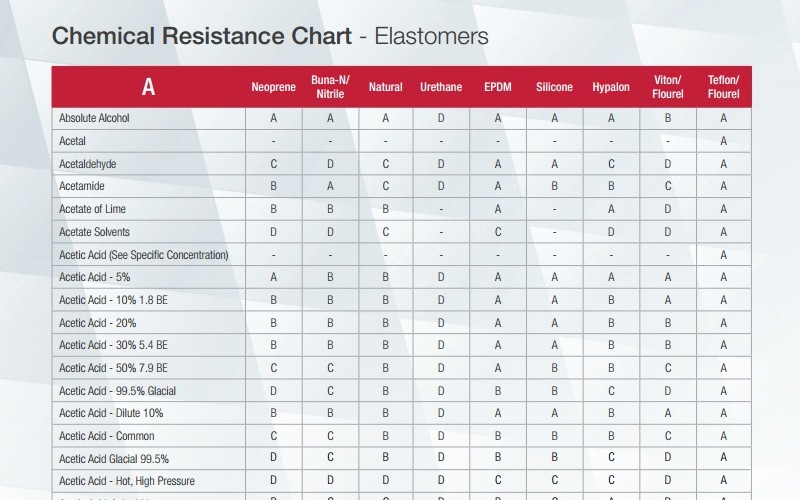
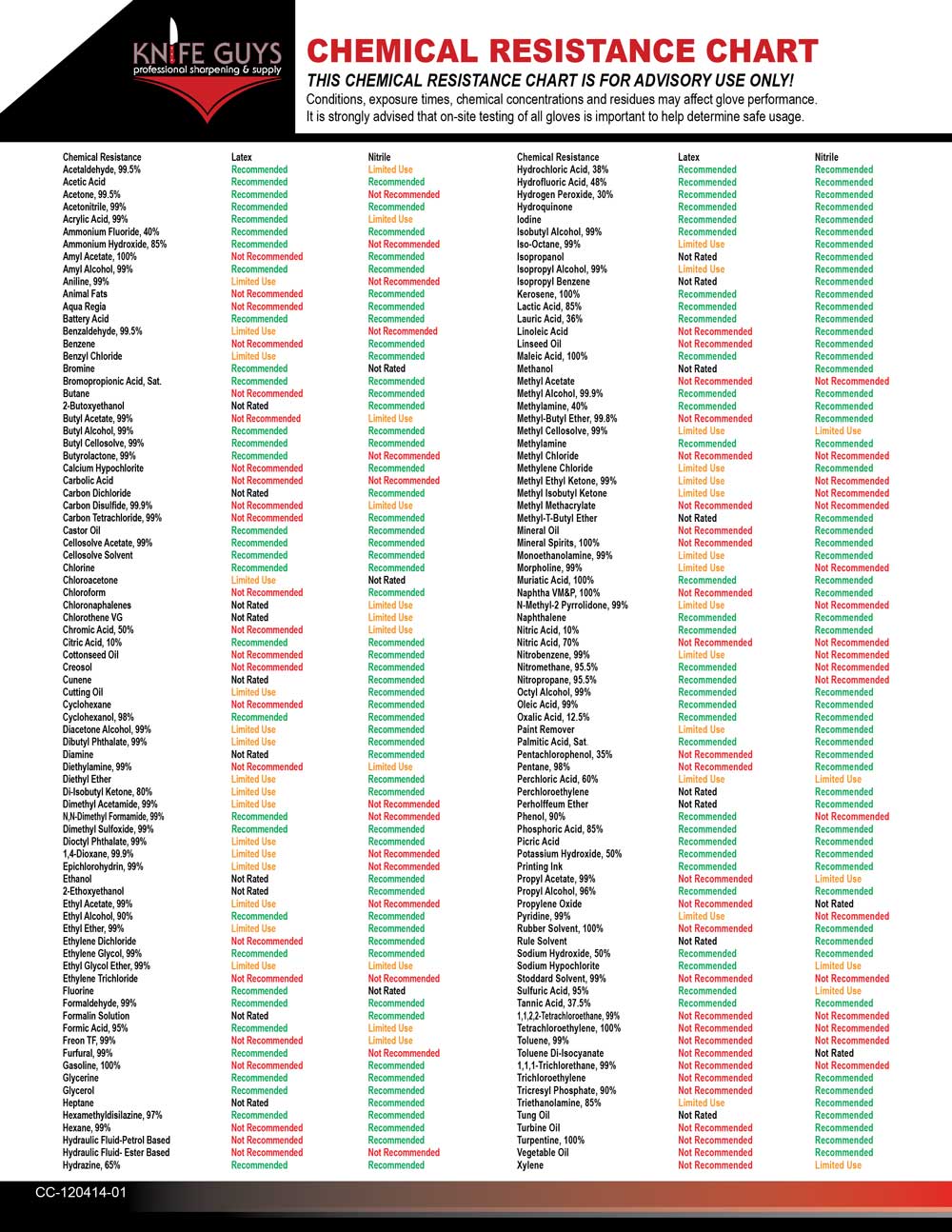
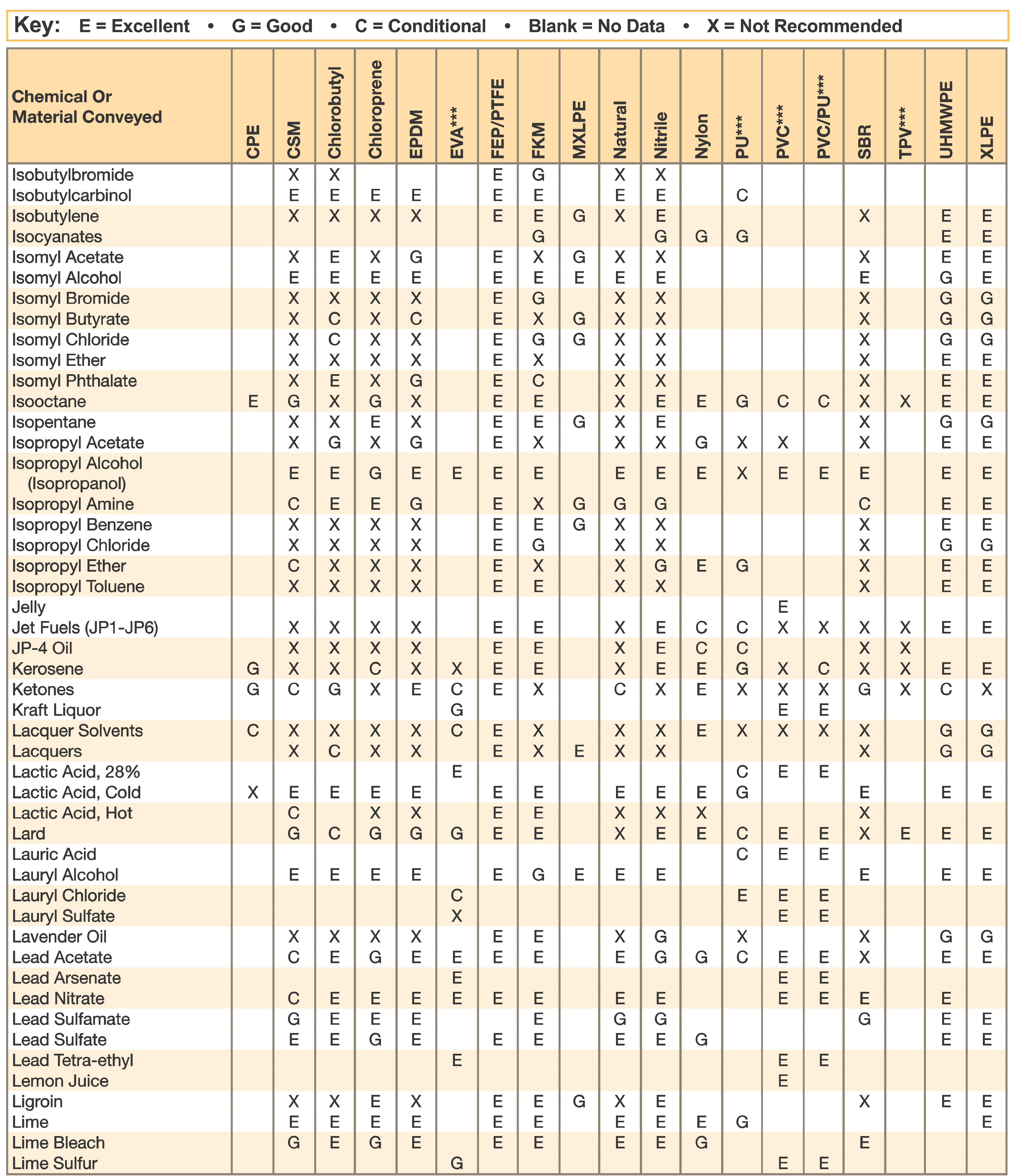
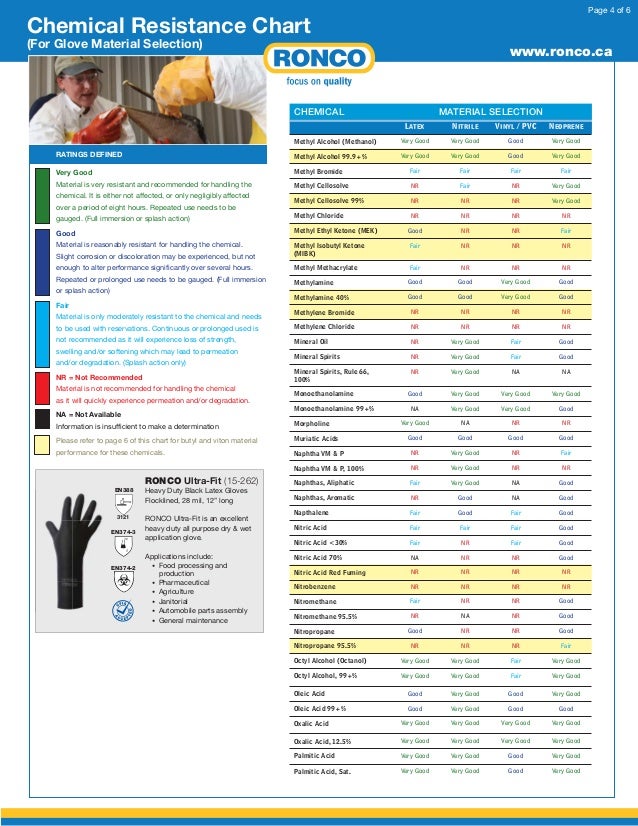
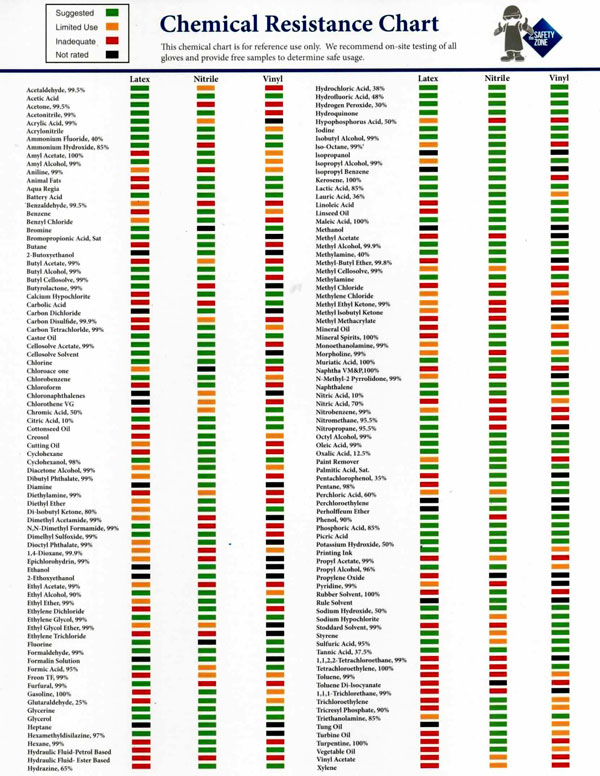
Nitrile Gloves Chemical Resistance Chart
Nitrile Gloves Chemical Resistance Chart - Identify the nitrile, the reagents, or both, needed to obtain a given carboxylic acid from a hydrolysis. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Based on the formulated ratio of acn and butadiene, the. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications. However, in the context of rubber, nitrile. Identify the products formed from the (acidic or basic) hydrolysis of a given nitrile. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance in various industrial applications. Nitrile, any of a class of organic compounds having molecular structures in which a cyano group (―c ≡ n) is attached to a carbon atom (c). It is a synthetic rubber, and is usually the ideal choice for any product that requires. The resulting material is appropriately. It is a synthetic rubber, and is usually the ideal choice for any product that requires. Based on the formulated ratio of acn and butadiene, the. Identify the products formed from the (acidic or basic) hydrolysis of a given nitrile. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications. Nitrile, any of a class of organic compounds having molecular structures in which a cyano group (―c ≡ n) is attached to a carbon atom (c). Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance in various industrial applications. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Identify the nitrile, the reagents, or both, needed to obtain a given carboxylic acid from a hydrolysis. However, in the context of rubber, nitrile. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Identify the products formed from the (acidic or basic) hydrolysis of a given nitrile. Based on the formulated ratio of acn and butadiene, the. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance. The resulting material is appropriately. It is a synthetic rubber, and is usually the ideal choice for any product that requires. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance in various industrial applications. Identify the nitrile, the reagents, or both, needed to obtain a given carboxylic acid from a hydrolysis. Nitriles. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Based on the formulated ratio of acn and butadiene, the. Nitriles are colourless solids or liquids with. The resulting material is appropriately. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications. It is a synthetic rubber, and is usually the ideal choice for any product that requires. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance in various industrial applications. Based on the formulated ratio of acn and butadiene, the. Nitrile, any of a class of organic compounds having molecular structures in which. Based on the formulated ratio of acn and butadiene, the. However, in the context of rubber, nitrile. Nitriles are colourless solids or liquids with. Identify the nitrile, the reagents, or both, needed to obtain a given carboxylic acid from a hydrolysis. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. The resulting material is appropriately. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. Nitriles are colourless solids or liquids with. Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. Discover the pros and. It is a synthetic rubber, and is usually the ideal choice for any product that requires. However, in the context of rubber, nitrile. Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn). Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications. However, in the context of rubber, nitrile. Identify the products formed from the (acidic or basic) hydrolysis. The resulting material is appropriately. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications. Nitriles are colourless solids or liquids with. Based on the formulated ratio of acn and butadiene, the. However, in the context of rubber, nitrile. Nitrile, in a broader sense, refers to any organic compound with a nitrogen atom that is connected to a carbon atom with a triple bond. The resulting material is appropriately. Nitrile is a synthetic rubber copolymer that is created when acrylonitrile (acn) and butadiene are bonded together using polymerization. It is a synthetic rubber, and is usually the ideal choice for any product that requires. Discover the pros and cons of nitrile rubber (nbr), including its oil resistance, temperature limits, and performance in various industrial applications. Nitrile, any of a class of organic compounds having molecular structures in which a cyano group (―c ≡ n) is attached to a carbon atom (c). Nitriles are colourless solids or liquids with. Identify the products formed from the (acidic or basic) hydrolysis of a given nitrile. Nitrile butadiene rubber (nbr), also known as nitrile rubber, is a synthetic rubber widely used in industrial applications.Nitrile Gloves Chemical Resistance Chart Cool Product Assessments, Promotions, and Buying Advice
Ansell Nitrile Gloves Chemical Resistance at Ruby Robinson blog
A Guide to Nitrile Gloves Chemical Resistance —
Chemical Resistance Chart Elastomers Phelps Industrial Products
Chemical Resistant Gloves Hand Protection uvex Safety Australia
Chemical Resistance Chart Gloves By Web
Nitrile Gloves Chemical Resistance Chart at Sharon Boyle blog
Chemical Compatibility Chart For Rubber at Jessie Thurman blog
Nitrile Gloves Compatibility Chart A Visual Reference of Charts Chart Master
Glove Chemical Compatibility Chart Images Gloves and Descriptions
However, In The Context Of Rubber, Nitrile.
Based On The Formulated Ratio Of Acn And Butadiene, The.
Identify The Nitrile, The Reagents, Or Both, Needed To Obtain A Given Carboxylic Acid From A Hydrolysis.
Related Post: