Anodization Chart
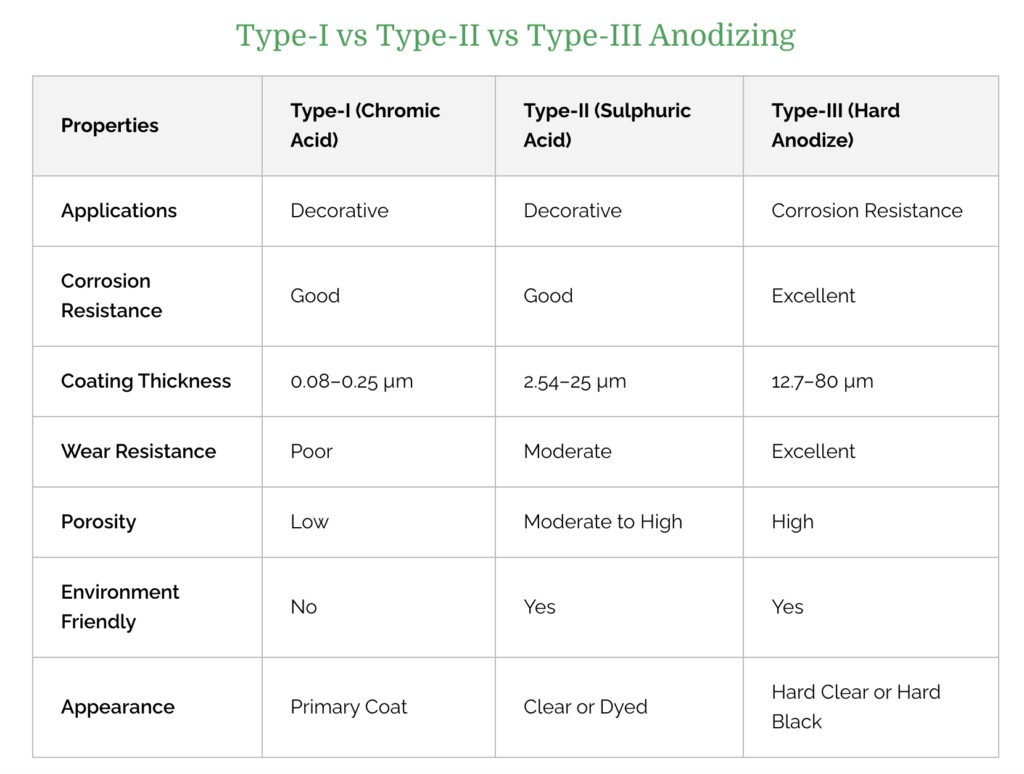
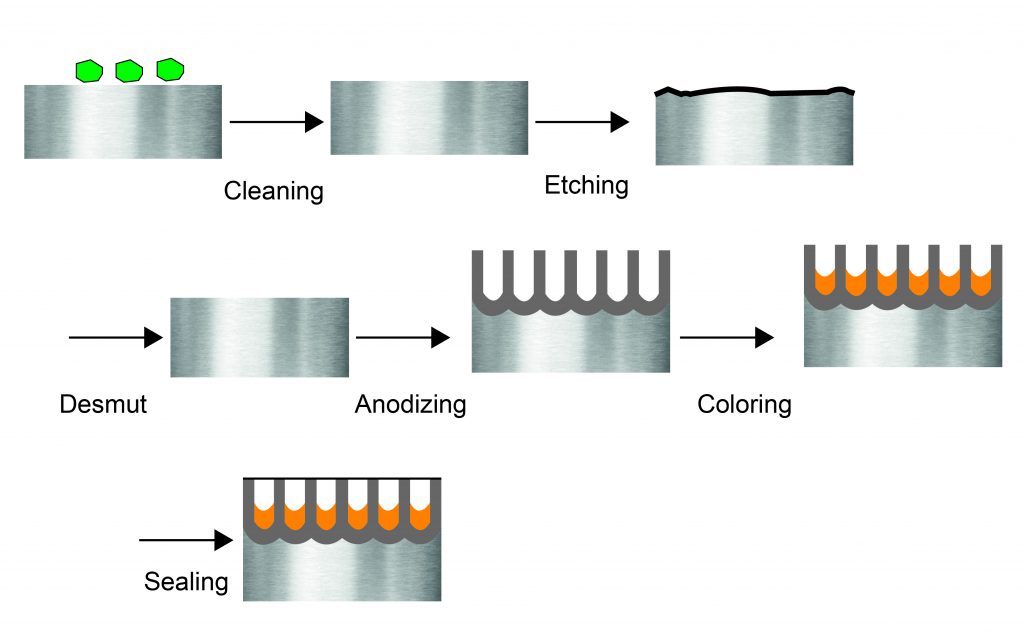
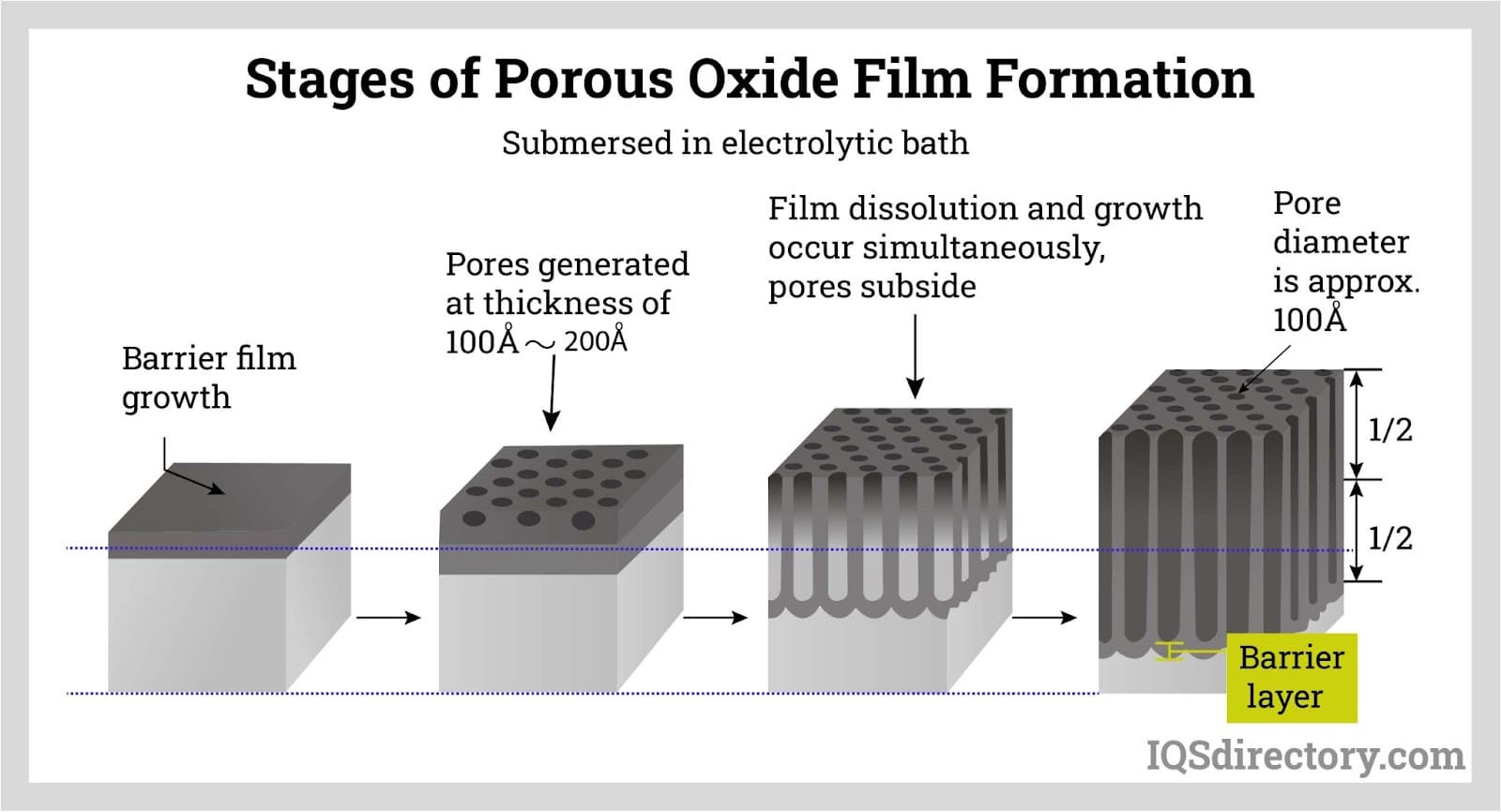
Anodization Chart - Aluminum is ideally suited to anodizing,. A limited number of metals, such as stainless steels,. The resulting finish, depending on the process, is the second hardest substance known. Anodizing consists of electrically depositing an oxide film from aqueous solution onto the surface of a metal, often aluminum, which serves as the anode in an electrolytic cell. Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. The process is called anodizing because the part to be. An electrochemical process that involves coating a metal part with an oxide surface layer, anodizing gives the part. It is an electrochemical process that thickens and toughens the naturally occurring protective oxide. Anodizing is a key step in manufacturing aluminum cnc machined parts. Anodization is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. What is anodizing and why is it important? Anodization is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. The resulting finish, depending on the process, is the second hardest substance known. Anodizing consists of electrically depositing an oxide film from aqueous solution onto the surface of a metal, often aluminum, which serves as the anode in an electrolytic cell. Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. Anodization converts traditional metal surfaces to a beautiful, durable, and high performing interior solution by implementing an accelerated. Anodizing is a key step in manufacturing aluminum cnc machined parts. Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. A limited number of metals, such as stainless steels,. It is an electrochemical process that thickens and toughens the naturally occurring protective oxide. What is anodizing and why is it important? The resulting finish, depending on the process, is the second hardest substance known. Anodization converts traditional metal surfaces to a beautiful, durable, and high performing interior solution by implementing an accelerated. Aluminum is ideally suited to anodizing,. An electrochemical process that involves coating a metal part with an oxide surface layer, anodizing. Anodization converts traditional metal surfaces to a beautiful, durable, and high performing interior solution by implementing an accelerated. It is an electrolytic process. The resulting finish, depending on the process, is the second hardest substance known. Anodizing is a key step in manufacturing aluminum cnc machined parts. Anodizing consists of electrically depositing an oxide film from aqueous solution onto the. Anodization converts traditional metal surfaces to a beautiful, durable, and high performing interior solution by implementing an accelerated. It is an electrolytic process. What is anodizing and why is it important? Aluminum is ideally suited to anodizing,. Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also. What is anodizing and why is it important? Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. The process is called anodizing because the part to be. The resulting finish, depending on the process, is the second hardest substance known. Anodizing is a key step. What is anodizing and why is it important? Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. It is an electrochemical process that thickens and toughens the naturally occurring protective oxide. A limited number of metals, such as stainless steels,. Aluminum is ideally suited to anodizing,. The resulting finish, depending on the process, is the second hardest substance known. What is anodizing and why is it important? Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. Aluminum is ideally suited to anodizing,. Anodizing consists of electrically depositing an oxide film from. What is anodizing and why is it important? Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. An electrochemical process that involves coating. Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. What is anodizing and why is it important? An electrochemical process that involves coating a metal part with an oxide surface layer, anodizing gives the part. Anodizing is a key step in manufacturing aluminum cnc machined parts. The. An electrochemical process that involves coating a metal part with an oxide surface layer, anodizing gives the part. The process is called anodizing because the part to be. What is anodizing and why is it important? A limited number of metals, such as stainless steels,. It is an electrolytic process. Aluminum is ideally suited to anodizing,. Anodizing consists of electrically depositing an oxide film from aqueous solution onto the surface of a metal, often aluminum, which serves as the anode in an electrolytic cell. It is an electrochemical process that thickens and toughens the naturally occurring protective oxide. The process is called anodizing because the part to be. It is. Anodizing is a key step in manufacturing aluminum cnc machined parts. Anodizing is an electrochemical technique that improves both the functionality and appearance of nonferrous metals, most commonly aluminum and its alloys, but also copper,. The resulting finish, depending on the process, is the second hardest substance known. An electrochemical process that involves coating a metal part with an oxide surface layer, anodizing gives the part. The process is called anodizing because the part to be. Anodization converts traditional metal surfaces to a beautiful, durable, and high performing interior solution by implementing an accelerated. It is an electrolytic process. It is an electrochemical process that thickens and toughens the naturally occurring protective oxide. Anodizing consists of electrically depositing an oxide film from aqueous solution onto the surface of a metal, often aluminum, which serves as the anode in an electrolytic cell. What is anodizing and why is it important? Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts.Your Basic Guide on How to Anodize Aluminum Parts rapiddirect
Us Anodizing Color Chart Paint Color Options Are Changing At
Materials Free FullText Progress in NanoEngineered Anodic Aluminum Oxide Membrane Development
Aluminum Anodizing Chart at Indiana Brownless blog
How to Anodize Aluminum Type II Anodizing for Beginners AluConsult
What Is Anodizing Process Types And Advantages Non Fe vrogue.co
Aluminum Anodizing Types at Barry Burgess blog
aluminum anodized color chart Anodized aluminum sheet colors for selection
Aluminum Anodizing Color Chart
Anodization Chart
Anodization Is An Electrolytic Passivation Process Used To Increase The Thickness Of The Natural Oxide Layer On The Surface Of Metal Parts.
A Limited Number Of Metals, Such As Stainless Steels,.
Aluminum Is Ideally Suited To Anodizing,.
Related Post:






 - WATERMARK.png/:/rs=w:1240,cg:true,m)